 
However, for an existing plant, the heat exchanger areas are fixed. This assumption is valid if we are designing a new plant and the equipment is being sized for construction. In that study, assuming variable area of the heat exchangers, we found that one percent liquid carry over can cause considerable change in compressor power requirement and heat exchanger duty. Due to the importance of separators, in the July Tip of the Month (TOTM), we studied the effect of liquid carry over in a simple dew point control plant. Many facility operating problems are related to improperly designed or under-sized gas-liquid separators. To learn more about similar cases and how to minimize operational problems, we suggest attending our G4 (Gas Conditioning and Processing) and G5 (Gas Conditioning and Processing – Special) courses. 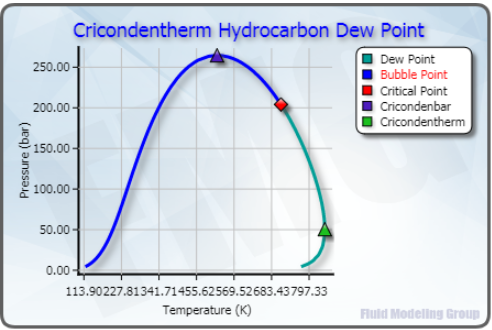
Figure 4 indicates that the hydrocarbon dew point and water dew point curves intersect at 100 ☏ and 1000 psia which are the conditions of the separator.ĭue to the fact that hydrate formation is controlled mostly by lighter components, there are only small variations of the hydrate formation curve and its inhibition by 25 weight percent methanol in all four mixtures. At this condition, the water content by SRK EoS is 0.0012 mole fraction equivalent of 57 lb m/MMSCF or 914 kg/10 6 std. Due to the removal of free water and heavy hydrocarbons from mixture 2, the phase envelope and the water dew point curve have moved to the left, as expected. So, below about 1400 psia, the wet hydrocarbon dew point is predicted while for pressures above 1400 psia, the water dew point (the higher one) is predicted.įinally, mixture 2 has been passed through a separator at 100 ☏ and 1000 psia and the resulting vapor compositions from a three-phase flash calculation based on the SRK EoS is shown in the last column of Table 1 as mixture 4. Also note that at a specified pressure, the higher of the two dew points (hydrocarbon and water) have been calculated by the SRK EoS. It is interesting to note that the dry hydrocarbon dew points and the wet hydrocarbon dew points predicted by SRK coincide very closely with each other the difference is practically negligible. The compositions of natural gases studied in this work are shown in Table 1. This version of SRK is tailor fitted for water-hydrocarbon systems. In this work we will use Figure 6.1 of Volume 1 and the modified Soave-Redlich-Kwong (SRK EoS) reported in GPA RR-42 by Erbar et al. The details of these methods can be found in Chapter 6 of Volume 1 and Chapter 9 of Volume 3 of “Gas Conditioning and Processing”. There are several methods of calculating of water content. The water content of a gas depends on the system temperature, pressure and composition of the water containing gas. What form/phase water is in at operating conditions, during start-up, during shut-down and during upsets?Ī phase envelope with hydrate and water dew point curves is an excellent tool to answer the above questions.For design and operation of a plant it is important to know: Once hydrates are formed, they are hard to remove. 
Hydrates can even form at warm temperatures in the presence of water. 
A question that comes to mind is: “why water is important?” The presence of water may cause corrosion, freezing and hydrate formation. In future tip, we will address wet sour gas. In this tip, we will demonstrate the water-sweet natural gas phase behavior. In the past Tips of the Month, we discussed the phase behavior of water-free natural gas mixtures. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
